 
Mendeleev was able to predict the existence of elements before they were discovered or synthesized by looking at gaps in his table. He put the elements into a grid with increasing atomic weights and noticed the elements in each column had similar chemical properties. 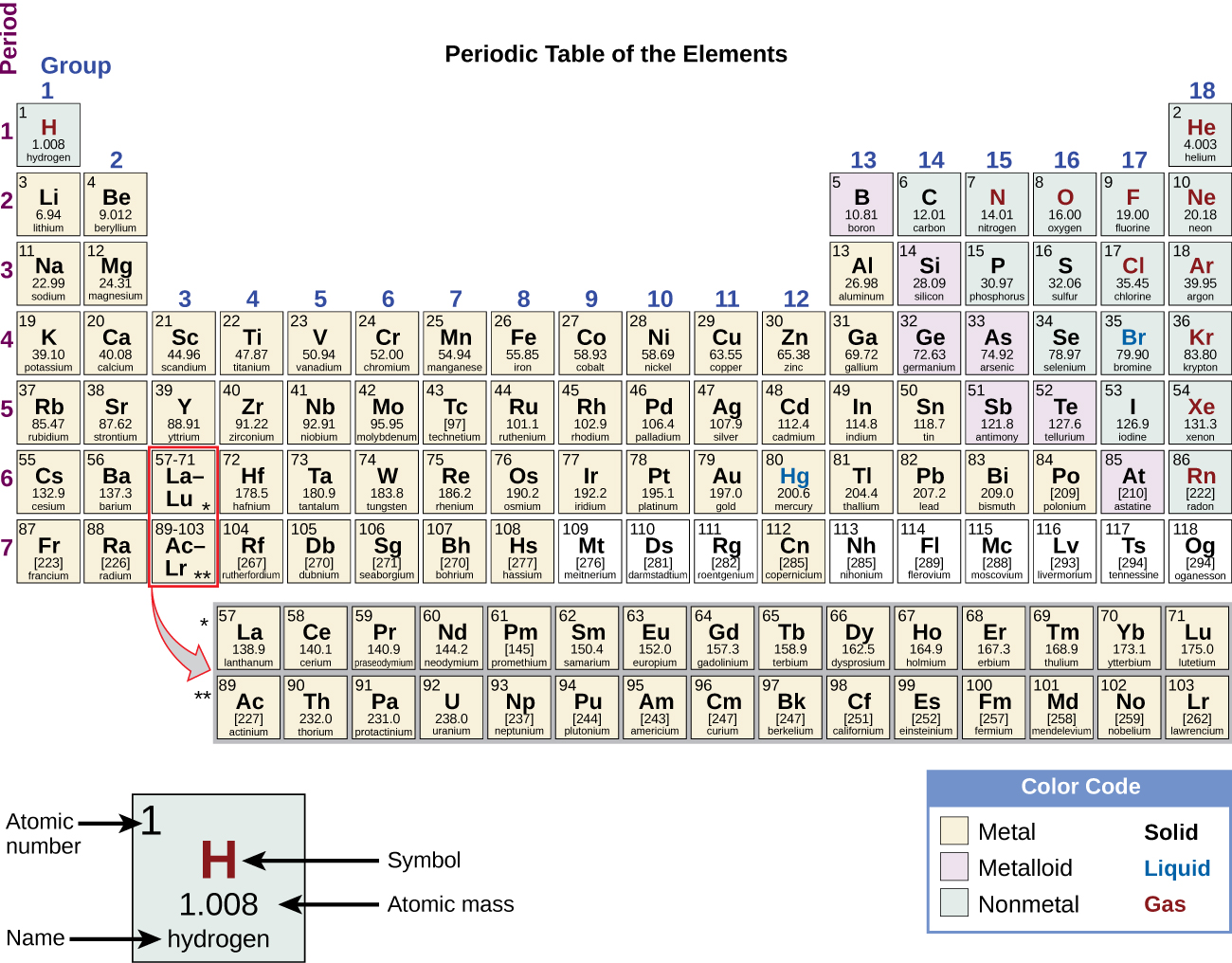
The first version was constructed by Dmitri Mendeleev in 1869. 
The periodic table is a way to organize the elements based on their similarities. The integer number in the box with the element symbol is the atomic number of the element and also the number of protons in each atom of the element. We number the elements, beginning with hydrogen, number one, in integers up to the largest number. As you read down the chart from top to bottom, a line of elements is a Group or Family. As you read across the chart from right to left, a line of elements is a Period. The Periodic Chart of the Elements is just a way to arrange the elements to show a large amount of information and organization. The Periodic Chart of Table of the Elements Overview
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
